Consent Form for COV-GEN Study (Adult Respondent)
PLEASE READ THE FOLLOWING CAREFULLY.
IF YOU HAVE ANY QUESTIONS, PLEASE LET THE RESEARCHER KNOW.
Study title
A COVID-19 patients’ Cohort: Genetic variants and other host risk factors associated with severity of infection, outcome and long-term complications (COV-GEN)
Introduction
You are invited to participate in this study. The following information outlines the details of this study, and we hope that it helps you decide on participation. Please read the information carefully. If you have any doubts or questions, please refer them to the researcher. Participation in this study is entirely voluntary.
Study objectives
Malaysia had its first case of COVID-19 in January 2020, and since then, the nation had seen three waves of the pandemic. Currently, we are facing the worst wave with daily cases between 6000-8000 patients and recently (in May 2021) scoring the highest in terms of the number of cases per million population, even ahead of India. During the 2nd wave, 90% of cases in Malaysia were mild, and we had a low case fatality rate. However, in recent weeks in May 2021, we have seen a higher proportion of moderate to severe cases and a much higher case fatality rate. The emergence of the COVID-19 variant of concern and variant of interest in Malaysia is likely to play a role in increased transmissibility and severity of the cases.
This study aims to identify the risk factors associated with the increased severity of COVID-19 and the Long Covid phenomenon involving our Malaysian patients. Additional research will help us confirm if these are risk factors for severe COVID-19 illness and determine if other factors increase a person’s risk. Using these data, we will develop a risk factor scoring system that can manage COVID-19 cases by reducing death cases and long-term complications.
This study involves COVID-19 patients of all categories (Category 1-5), including children and teenagers. This study requires a repeat of data collection at several time points, when: (1) one is admitted at ward/quarantine centre; (2) one is recovered and discharged from ward; (3) 3 months, (4) 6 months, and (5) 12 months after COVID-19 recovery. During the data collection, the participant will be asked to fill in the questionnaire via an online platform. As many as 20 ml of blood and 10 ml of urine will be requested from the participants. The blood and urine samples are essential for studying the host-COVID-19 relationship.
The following diagram summarises the flow of this study:
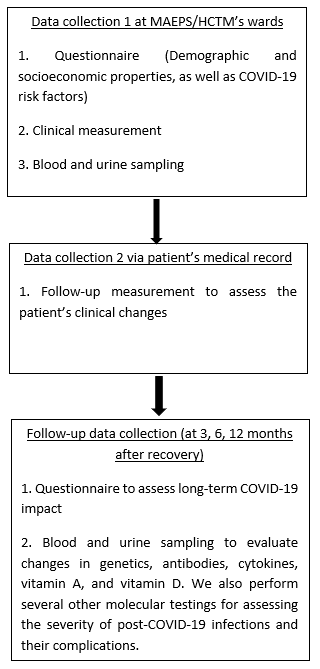 Figure 1
Figure 1

Figure 2
Inclusion Criteria as COVID-19 Patients
Patients (from category 1 to 5) who are diagnosed as positive for COVID-19 infection and fulfil clinical diagnostic criteria.
- COVID-19 survivors recovered and were discharged within 3 months.
- Only COVID-19 survivors who reside in Kuala Lumpur and Selangor will be included in this study to ease the data collection at a later phase.
Inclusion Criteria as Control Cases
- Patients who are negative for COVID-19 infection and are healthy
- Patients who were negative for COVID-19 screening but were admitted to the hospital due to lung infections.
Exclusion Criteria for this Study
- Participants who do not agree or wish to withdraw their participation at any time point during the study.
What should you do?
If you agree to participate in this study, you need to do the following:
- Answer all questions in the questionnaire (this Google Form) independently. If you need any assistance, please let the researcher know.
- As many as 20 ml of blood will be drawn from your vein. The medical doctor will draw your blood using a needle-sized 23-24 mm, and you might feel pain.
- As many as 10 ml of urine will be requested from you. You will need to urinate into the container given to you.
- You are agreed for: a follow-up data collection, blood and urine sampling, and our access to your health record.
Blood and urine collected were used to assess changes in genetics, antibodies, cytokines, vitamin A, and vitamin D. We also perform several other molecular testings to evaluate the severity of post-COVID-19 infections and their complications.
Benefits and Risks of Participating in this Study
There are no known severe side effects in this study because all the procedures are routine laboratory testings for diseases in a research setting. The only risk is you will feel the pain when drawing blood.
This study provides direct benefits to the participants as they will better understand their COVID-19 disease status. The participants will also learn about the host risk factors and complications after COVID-19 infections. If the participants would like to request a copy of their blood and urine results, we (the researchers) will give it to you via email or phone calls based on the contact information collected in this questionnaire.
Do you need to Participate in this Study?
Your participation in this study is entirely voluntary, and you have the right to choose to participate or not. If you agree to participate in this study, we would like you to sign this consent form, proving that you are well-informed and understand the details regarding this study. By signing this consent form, you are not waiving any legal rights you have as a participant in this study. If you want to withdraw from this study later, your action will not affect any of the medical rights and privileges you are entitled to. The researcher also can refuse/reject your participation in this study due to specific reasons. If rejection happens, your rights as a patient will not be affected.
Data Confidentiality and Participant’s Identity
All information provided in this questionnaire and through our discussion is confidential. Respondent’s identity will be concealed/anonymised, and the data will be reported/presented as a group in the future.
Payment and Token of Appreciation
All laboratory testings that were performed in this study are free of charge. If you participated in this study, you would not be charged and paid.
Who to contact to learn more about this study?
If you have any additional questions regarding this study, please contact our lead researcher:
PROFESOR DATUK DR. A RAHMAN A JAMAL
Lead Researcher of COV-GEN Project
UKM Medical Molecular Biology Institute, Universiti Kebangsaan Malaysia,
Jalan Yaacob Latiff, Bandar Tun Razak, 56000 Cheras, Kuala Lumpur, Malaysia
E-mail: rahmanj@ppukm.ukm.edu.my
Tel: +603-91456321/+603-91717185

