Vancomycin Intermediate Resistance in S. aureus: Importance of the arginine deiminase (ADI) Pathway
By: Xin Ee Tan, Ph.D. and Hui-min Neoh, Ph.D.
Staphylococcus aureus is one of the most common Gram positive bacteria known to cause a wide range of infections in humans and to rapidly develop resistance towards clinically-introduced antibiotics. This is demonstrated by the report on the first penicillin-resistant S. aureus in 1942 right after the introduction of this drug in the early 1940’s, and the description of the first methicillin-resistant S. aureus (MRSA) in 1961, barely 2 years after the drug was used. The development of resistance by S. aureus towards virtually every available antibiotic had then increased the importance of vancomycin, a glycopeptide, as the ultimum refugium (last possible treatment).
In 1996, the first clinical isolate of vancomycin-intermediate S. aureus (VISA), Mu50, was reported from a surgical wound infection of a 4-month-old male infant in Juntendo University Hospital, Japan. Infections with VISA were subsequently identified from other parts of the world, including United States, France, South Korea, and Brazil. The successive emergence of S. aureus strains with decreased susceptibility to vancomycin therapy indicates that this drug is beginning to lose its clinical applicability clinically. Hence, various studies were, and are now, being carried out to obtain a better understanding of the underlying mechanisms of resistance as well as to discover potential targets for new antimicrobial chemotherapy.
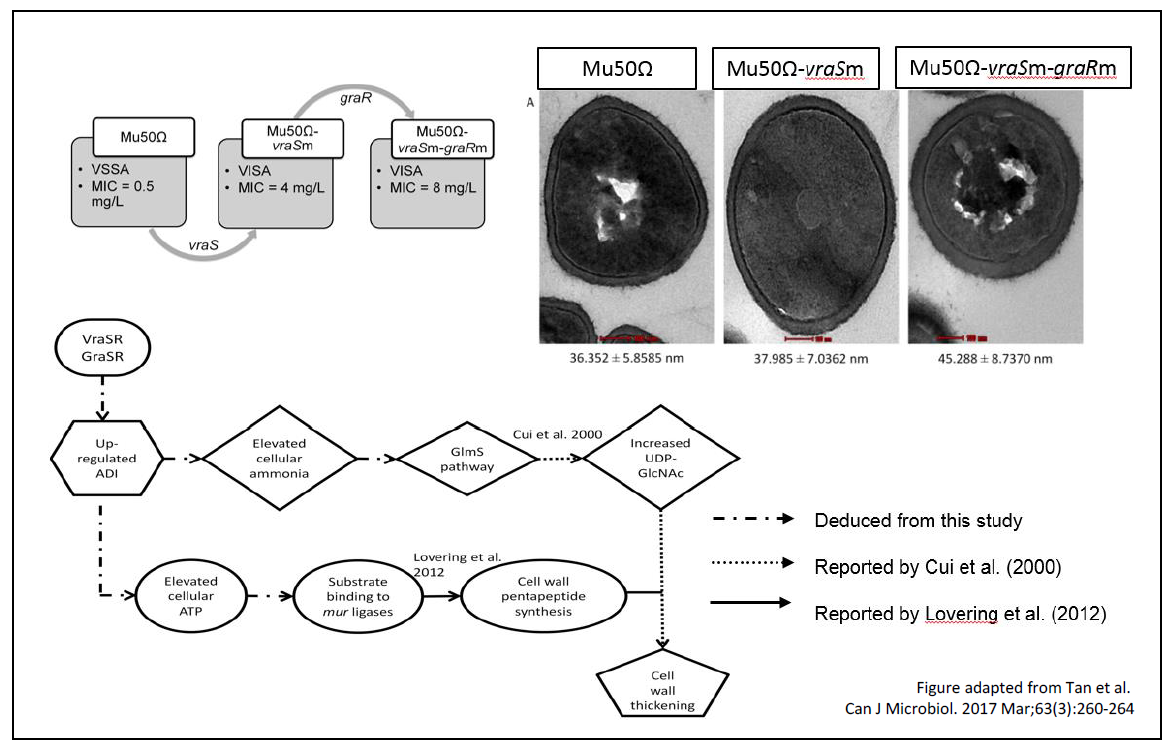
Previous studies revealed vraSR and graSR as 2 two-component regulatory genes associated with VISA. Mutations in these genes (vraS, T→A at position 700; and graR, A→G at position 590) are associated with vancomycin intermediate resistance. However, it remains unclear how these mutations contribute to resistance. In UMBI, we employed a proteomic approach to derive possible vraSR/graSR-associated vancomycin intermediate resistance regulatory pathway(s). Total proteins were extracted from a clinical vancomycin-susceptible S. aureus strain (Mu50Ω) and its genetically engineered VISA isogenic strains harbouring either only the vraS TàA mutation (strain Mu50Ω-vraSm) or both vraS T→A/graR A→G mutations (strain Mu50Ω-vraSm-graRm). Comparative proteomic profiling of the 3 isogenic strains revealed up-regulated ArcB of the arginine deiminase (ADI) pathway, where validation experiments showed elevation of ammonia and ATP molecules in concert with thickened cell walls of VISAs, indicating involvement of ADI pathway in the generation of vraS/graR mutation-associated VISAs.
Read more about this study at Tan et al., Malaysian Journal of Microbiology, 2016, 12(6): 498-505 (https://www.researchgate.net/publication/312159813_Comparative_proteomics_profiling_reveals_down regulation_of_Staphylococcus_aureus_virulence_in_achieving_intermediate_vancomycin_resistance) and
Tan et al., Canadian Journal of Microbiology,2017,63(3):260-264
(https://www.researchgate.net/publication/309301724_Activated_ADI_pathway_the_initiator_of_intermediate_vancomycin_resistance_in_Staphylococcus_aureus)
By: Xin Ee Tan, Ph.D. and Hui-min Neoh, Ph.D.